Popular Examples
- Resumes
- Cover Letters
- CVs
- Idioms
- Excel Templates
- Business Examples
- Business Letters
- Love Letters
- Personal Letters
- Science
- Math Examples
More Science
- Example of Parasitism
- Example of Phenotype
- Example of Photosynthesis
- Example of Physical Change
- Example of Physical Property
- Example of Physical Weathering
- Example of Pitch
- Example of Pivot Joint
- Example of Plantae
- Example of Plasma
- Example of Pseudoscience
- Example of Radiant Energy
- Example of Radiation
- Example of Rube Goldberg Machine
- Example of Sedimentary Rock
- Example of Solution
- Example of Speciation
- Example of Species
- Example of Electrolyte
- Example of Gametes
- Example of Gametic Isolation
- Example of Gamma Rays
- Example of Gas
- Example of Gas to Solid
Examples of Single Replacement
|
This is a fairly broad term used in relation to oxidization. The actual process refers to an oxidization/reduction chemical reaction which occurs when an ion moves out of one compound and into another. What actually happens is that the nature of the compound is changed. Elements in the compound may not transfer evenly, which results in a different expression of the single replacement, as below: |
Example of Single Replacement:
|
X + YZ = Y+XZ 2AB + C = 2A = CB2 (This expression may occur with additional modifiers created by the presence of compounds like O3, or other structures. https://en.wikipedia.org/wiki/Single_displacement_reaction https://dl.clackamas.edu/ch104-04/single.htm https://homepage.smc.edu/walker_muriel/single_replacement_battery_lab_procedure.htm |
Image Example of Single Replacement:
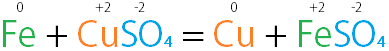 A single displacement reaction example. |