Popular Examples
- Resumes
- Cover Letters
- CVs
- Idioms
- Excel Templates
- Business Examples
- Business Letters
- Love Letters
- Personal Letters
- Science
- Math Examples
More Science
- Example of Parasitism
- Example of Phenotype
- Example of Photosynthesis
- Example of Physical Change
- Example of Physical Property
- Example of Physical Weathering
- Example of Pitch
- Example of Pivot Joint
- Example of Plantae
- Example of Plasma
- Example of Pseudoscience
- Example of Radiant Energy
- Example of Radiation
- Example of Rube Goldberg Machine
- Example of Sedimentary Rock
- Example of Solution
- Example of Speciation
- Example of Species
- Example of Electrolyte
- Example of Gametes
- Example of Gametic Isolation
- Example of Gamma Rays
- Example of Gas
- Example of Gas to Solid
Example of Ionic Crystal
|
Ionic crystals are compounds of elements held together by ionic bonds. The compound must have at least two components which are ionized, although many have several. Bonds are quite strong, usually electrostatic, at 1000kj per molecule. There are no free electrons. As crystals, they from regulated shapes either simple cubes or complex forms including the poly faceted forms of tetrahedral and octahedral forms. The more complex types of compounds are those with multiple ionic elements as components. Ionic crystals are generally lattice structures, very hard and with high melting points. These substances are also known as ionic solids. |
Examples of Ionic Crystals:
|
Salt Potassium Chloride Lead sulphate Magnesium oxide Iron oxide Calcium Titanium oxide Magnetite Chromite https://www.seas.upenn.edu/~chem101/sschem/ionicsolids.html https://ocw.mit.edu/NR/rdonlyres/1498B922-14DD-4D52-903D-874D18F14C07/0/lecture05_slides.pdf https://en.wikipedia.org/wiki/Ionic_crystal |
Image Example of Ionic Crystal:
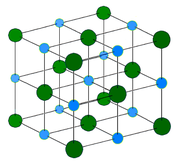 Sodium chloride (halite) crystal structure. Each atom has six nearest neighbors, with octahedral geometry. This arrangement is known as cubic close packed (ccp). Light blue = Na+ Dark green = Cl- |