Popular Examples
- Resumes
- Cover Letters
- CVs
- Idioms
- Excel Templates
- Business Examples
- Business Letters
- Love Letters
- Personal Letters
- Science
- Math Examples
More Science
- Example of Parasitism
- Example of Phenotype
- Example of Photosynthesis
- Example of Physical Change
- Example of Physical Property
- Example of Physical Weathering
- Example of Pitch
- Example of Pivot Joint
- Example of Plantae
- Example of Plasma
- Example of Pseudoscience
- Example of Radiant Energy
- Example of Radiation
- Example of Rube Goldberg Machine
- Example of Sedimentary Rock
- Example of Solution
- Example of Speciation
- Example of Species
- Example of Electrolyte
- Example of Gametes
- Example of Gametic Isolation
- Example of Gamma Rays
- Example of Gas
- Example of Gas to Solid
Example of Ionic Bond
An ionic bond is a strong bond created by combination of a metal and a non metal. The materials join through electrostatic attraction between positive and negative charges. The metal provides electrons, creating a stable cation configuration, with a reciprocal anion configuration in the non metal. In effect the two materials lock into a mutually supportive configuration as a compound. |
Examples of Ionic Bond:
|
Salt is the classic ionic bond. Sodium, the metal, and chlorine, the halide, combine to form a strong compound. https://en.wikipedia.org/wiki/Ionic_bond https://simple.wikipedia.org/wiki/Ionic_bond https://www.elmhurst.edu/~chm/vchembook/143Aioniccpds.html |
Image Example of Ionic Bond:
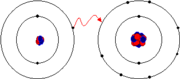 Electron configurations of lithium and fluorine. Lithium has one electron in its outer shell, held rather loosely because the ionization energy is low. Fluorine carries 7 electrons in its outer shell. When one electron moves from lithium to fluorine, each ion acquires the noble gas configuration. The bonding energy from the electrostatic attraction of the two oppositely-charged ions has a large enough negative value that the overall bonded state energy is lower than the unbonded state. |