Popular Examples
- Resumes
- Cover Letters
- CVs
- Idioms
- Excel Templates
- Business Examples
- Business Letters
- Love Letters
- Personal Letters
- Science
- Math Examples
More Science
- Example of Parasitism
- Example of Phenotype
- Example of Photosynthesis
- Example of Physical Change
- Example of Physical Property
- Example of Physical Weathering
- Example of Pitch
- Example of Pivot Joint
- Example of Plantae
- Example of Plasma
- Example of Pseudoscience
- Example of Radiant Energy
- Example of Radiation
- Example of Rube Goldberg Machine
- Example of Sedimentary Rock
- Example of Solution
- Example of Speciation
- Example of Species
- Example of Electrolyte
- Example of Gametes
- Example of Gametic Isolation
- Example of Gamma Rays
- Example of Gas
- Example of Gas to Solid
Example of Ion
An ion is an atom or group of atoms with an unequal number of protons and electrons, meaning the atom(s) become positively or negatively charged. Ions are a common part of chemical and atomic processes, providing bonding and interactions to form compounds as positive and negative forces interact. Various types of ions exist for all elements, affecting their natural properties.the generic term ion covers both types of ion. A negatively charged ion, having more electrons than protons is called anion. |
Examples of Ion:
|
These are some of the essential ions humans require for their daily lives: Sodium chloride (salt)Magnesium phosphate Calcium phosphate Sodium sulphate https://simple.wikipedia.org/wiki/Ion https://en.wikipedia.org/wiki/Ion |
Image Example of Ion:
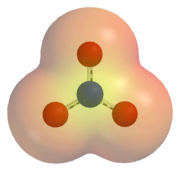 An electrostatic potential map of the nitrate ion. Areas coloured red are lower in energy than areas colored yellow. |